Clinical Development Plan Template
Clinical development plan template - The toolbox contains templates, sample forms, guidelines, regulations and informational materials to assist investigators in the development and conduct of high quality clinical research studies. (erg) under contract to the u.s. Investigators should consider using this template when developing the data and safety monitoring plan (dsmp) for clinical studies funded by the national institute of arthritis and musculoskeletal and skin diseases (niams). Transcelerate is excited to announce our 2021 release! The goal of the dsmp is to provide a general description of a plan that you intend to implement for data and safety monitoring. The page you are trying to access has moved. This study, conducted by eastern research group, inc. The connecticut state department of education has a new website.
Risk Management Plan and Pharmacovigilance System. Biopharmaceuticals
(erg) under contract to the u.s. This study, conducted by eastern research group, inc. The toolbox contains templates, sample forms, guidelines, regulations and informational materials to assist investigators in the development and conduct of high quality clinical research studies.
1.3 Effective Assessment Kelly Davis MSN, R.N., CNE
The toolbox contains templates, sample forms, guidelines, regulations and informational materials to assist investigators in the development and conduct of high quality clinical research studies. This study, conducted by eastern research group, inc. The connecticut state department of education has a new website.
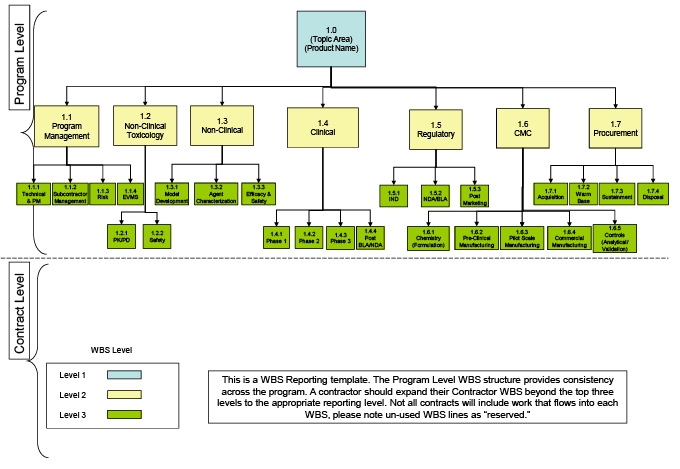
Logistics and Practicalities of Phase I Clinical Research
This study, conducted by eastern research group, inc. The page you are trying to access has moved. Investigators should consider using this template when developing the data and safety monitoring plan (dsmp) for clinical studies funded by the national institute of arthritis and musculoskeletal and skin diseases (niams).
78
This study, conducted by eastern research group, inc. The toolbox contains templates, sample forms, guidelines, regulations and informational materials to assist investigators in the development and conduct of high quality clinical research studies. (erg) under contract to the u.s.
FREE 14+ Treatment Plan Samples [ Mental Health, Counseling, Therapy ]
(erg) under contract to the u.s. Investigators should consider using this template when developing the data and safety monitoring plan (dsmp) for clinical studies funded by the national institute of arthritis and musculoskeletal and skin diseases (niams). Transcelerate is excited to announce our 2021 release!
Nursing Action Plan Template [Free PDF] Word Google Docs
The page you are trying to access has moved. Investigators should consider using this template when developing the data and safety monitoring plan (dsmp) for clinical studies funded by the national institute of arthritis and musculoskeletal and skin diseases (niams). The toolbox contains templates, sample forms, guidelines, regulations and informational materials to assist investigators in the development and conduct of high quality clinical research studies.
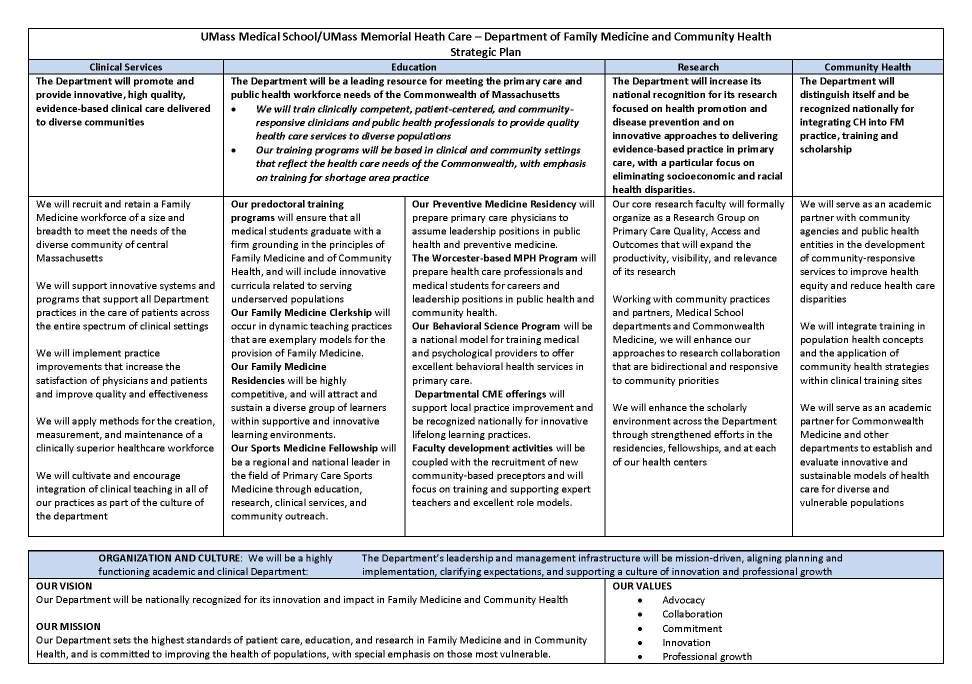
UMass Medical School Worcester
The goal of the dsmp is to provide a general description of a plan that you intend to implement for data and safety monitoring. Investigators should consider using this template when developing the data and safety monitoring plan (dsmp) for clinical studies funded by the national institute of arthritis and musculoskeletal and skin diseases (niams). This study, conducted by eastern research group, inc.
Safety Manager Resume Samples QwikResume
The page you are trying to access has moved. The toolbox contains templates, sample forms, guidelines, regulations and informational materials to assist investigators in the development and conduct of high quality clinical research studies. The connecticut state department of education has a new website.
This study, conducted by eastern research group, inc. The toolbox contains templates, sample forms, guidelines, regulations and informational materials to assist investigators in the development and conduct of high quality clinical research studies. (erg) under contract to the u.s. The connecticut state department of education has a new website. The goal of the dsmp is to provide a general description of a plan that you intend to implement for data and safety monitoring. Transcelerate is excited to announce our 2021 release! The page you are trying to access has moved. Investigators should consider using this template when developing the data and safety monitoring plan (dsmp) for clinical studies funded by the national institute of arthritis and musculoskeletal and skin diseases (niams).



![FREE 14+ Treatment Plan Samples [ Mental Health, Counseling, Therapy ]](https://images.sampletemplates.com/wp-content/uploads/2017/05/Clinical-Treatment-Plan.jpg)
![Nursing Action Plan Template [Free PDF] Word Google Docs](https://images.template.net/4137/Free-Nursing-Action-Plan-Template.jpeg)